Phytoplankton form the base of the marine food web. These microscopic, single-celled organisms float in seawater, taking in carbon dioxide and using light energy to make carbohydrates. Like land plants, phytoplankton need other elements and compounds (fertilizer) to perform photosynthesis in order to survive and thrive: Nitrogen is one of these key ingredients for phytoplankton growth.
Nitrogen is fascinating and somewhat unique because it cycles through many oxygenation states. This means that there is plenty of energy for organisms to harness and the nitrogen cycle can be used as a lens to understand microbial communities. The quantity of “fixed nitrogen” in the ocean, usually nitrate (NO3–) and ammonium (NH4+), is critical for the existence and development of phytoplankton, and plays a role in the biological carbon pump sequestering carbon dioxide from the atmosphere.
Being able to recognize the role of fixed nitrogen in ocean processes is important for understanding low-oxygen areas in the world’s oceans. Insight into microbial interactions in oxygen deficient waters will allow researchers to better predict the marine response to increased nutrient runoff, eutrophication, and hypoxia – all of which currently threaten the livelihoods of many coastal communities as a warming climate leads to the expansion of low oxygen “dead” zones.
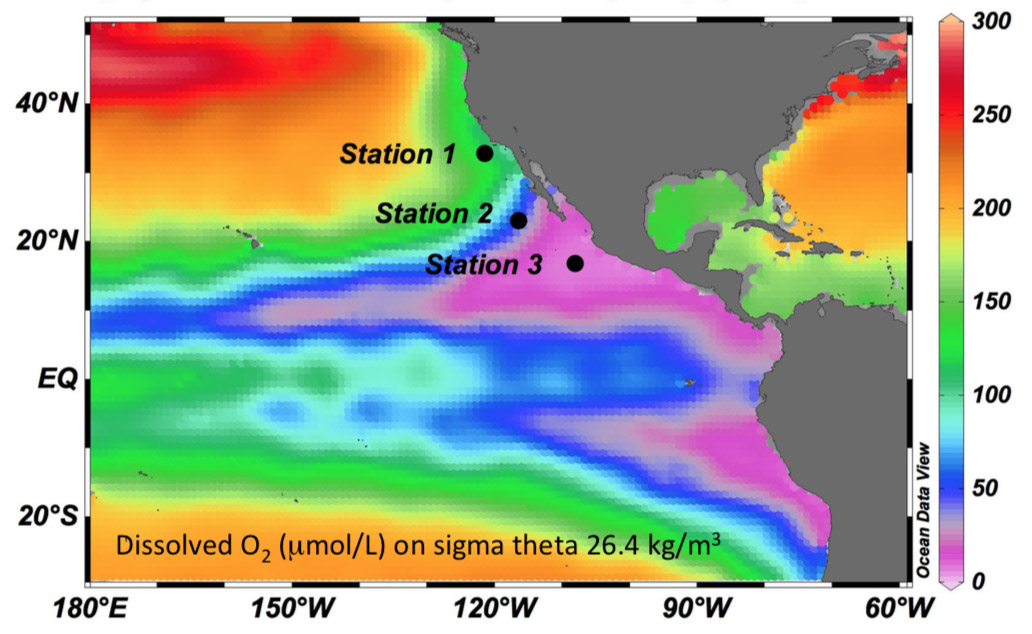
Investigating Oxygen Deficient Zones
Oxygen Deficient Zones (ODZs) are critical for the carbon, nitrogen, and energy balances in the ocean. In order to examine this on a detailed scale, scientists need to collect water samples that contain these compounds. However, it is difficult to sample, move, and incubate seawater from ODZs without oxygen contamination – until now. During a three week expedition aboard Falkor, to one of the largest ODZs in the Eastern Tropical North Pacific, Professor Karen Casciotti, Stanford University, and Professor Andrew Babbin, Massachusetts Institute of Technology, will attempt to find a solution with new technology.
Collaboration between oceanographers, chemists, microbial experts, and mechanical engineers will allow the team to develop technology that autonomously incubates and documents microbial processes. These measurements are traditionally conducted shipboard using water samples that are placed in simulated conditions to where they were obtained. The team aboard Falkor will assess the new device’s performance against traditional methods, with state of the art incubations on the ship to determine accuracy, and measure the rates of nitrogen transformation in the region.
In-Water Incubation Devices – A Good Thing
A new generation of autonomous in situ incubation devices that take measurements and record data will be tested and modified by the science team. Prototyped by McLane Research and in collaboration with Dr. Craig Taylor and Dr. Ginny Edgcomb, Woods Hole Oceanographic Institution, the new devices have been designed to autonomously fill an incubation chamber with seawater, conduct experimental treatments, and subsample the incubation at set intervals. Only a few tests have been able to capture water and incubate it at the same depth, and rarely in ODZs.
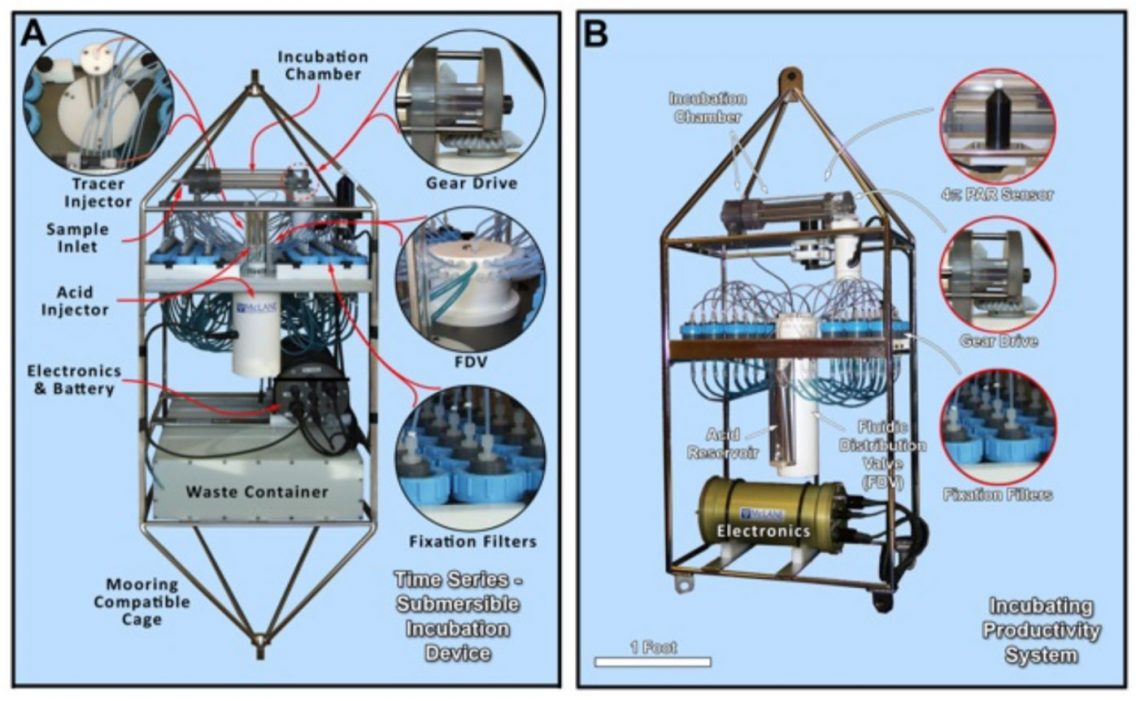
The team anticipates that oxygen will be leached out of the incubator when first deployed in the ODZ, so that after an initial “soak”, the scientists will be able to initiate experiments in truly oxygen-free conditions. The system can then be used to measure the rates of Nitrogen cycle processes at a variety of depths and corresponding levels of oxygen, nitrite, and ammonium in the water column.
By creating devices that carry out instructions underwater, efficiency will be increased, while preventing topside atmospheric contamination of samples. It will also help scientists to understand altered nitrogen content in a changing ocean and answer fundamental questions about microbes in these regions. Once refined, this technology will be applicable to a wide variety of important questions in marine science, and has the potential to be an irreplaceable tool in future studies.
Data & Publications
The resulting shipboard dataset can be accessed at Rolling Deck to Repository.
ADCP data is curated and processed by University of Hawaii.
Iodine Speciation measurements can be accessed in BCO-DMO.
An atlas of depth-gridded and density-gridded interpolated and un-interpolated Oxygen Deficient Zones in the Eastern tropical and subtropical Pacific Ocean has been published.
Bottle data and Chemical Analysis can be accessed through BCO-DMO.
- Babbin, A. (2019) Direct Measurements of Microbial Nitrogen Transformations in Suboxic Waters. Oral Presentation at Gordon Research Conference in Chemical Oceanography, Holderness, NH, USA.
- Evans, N., Boles, E., Kwiecinksi, J., Mullen, S., Wolf, M., Devol, A. et. al. (2020). The role of water masses in shaping the distribution of redox active compounds in the Eastern Tropical North Pacific oxygen deficient zone and influencing low oxygen concentrations in the eastern Pacific Ocean. Limn. and Ocean. 65(8), doi: 10.1002/lno.11412.
- Hardisty, D., Horner, T., Evans, N., Moriyasu, R., Babbin, A., Wankel, S., et. al. (2021). Limited iodate reduction in shipboard seawater incubations from the Eastern Tropical North Pacific oxygen deficient zone. Earth and Planetary Sci. Letters, Vol. 554, doi: 10.1016/j.epsl.2020.116676.
- Moriyasu, R., Evans, N., Bolster, K., Hardisty, D., and Moffett, J. (2020). The Distribution and Redox Speciation of Iodine in the Eastern Tropical North Pacific Ocean. Global Biogeochemical Cycles, 34(2), doi: 10.1029/2019GB006302.
- Wolf, M., Goodell, M., Dong, E., Dove, L., Zhang, C., Franco, L., et. al. (2020). A link between the ice nucleation activity and the biogeochemistry of seawater. Atmos. Chem. and Physics, 20(23), doi: 10.5194-acp-20-15341-2020. [This article is published as OPEN ACCESS].
- Cinay, T., Dumit, D., Woosley, R., Boles, E., Kwiecinski, J., Mullen, S., et al. (2022). Coincident Biogenic Nitrate and pH Maxima Arise in the Upper Anoxic Layer in the Eastern Tropical North Pacific. Global Biogeochemical Cycles, doi: 10.1029/2022GB007470. [This article has been published as OPEN ACCESS].
In the News
Solving Microbial Mysteries with Autonomous Technology
PAOC Spotlights • July 2nd, 2018
Monitoring and Tracking Ocean Microbes with LRAUVs
Environmental Monitor Magazine • Summer 2018