Expedition dates: April 12 – May 15, 2024
Oxygen Minimum Zones, or OMZs, are areas in the water column with less oxygen, primarily due to naturally occurring physical and biological processes within the Ocean. The core of the OMZs is anoxic, making them inhospitable to most large animals, yet microbes have adapted to live and thrive in this oxygen-starved environment. Microscopic organisms living in OMZs are diverse and immense, yet the data on their life cycles and the role they play in the Ocean’s biogeochemistry is scarce. Understanding the functions and activities of microbial communities in these environments is critical, as OMZs are thought to be growing due to climate change.
To characterize the microorganisms living in OMZs, Dr. Maria Pachiadaki of the Woods Hole Oceanographic Institution, U.S., has assembled an international team, including a team of Chilean scientists led by Dr. Osvaldo Ulloa of the University of Conceptión, to test new instruments in the Eastern Tropical Pacific Oxygen Minimum Zone. The central piece of equipment the team will use is the Submersible Incubation Device, or SID. This automated mini-laboratory collects and processes seawater samples in situ at specified depths to study biogeochemical processes while accurately measuring trace amounts of oxygen in the water. The team will also use a microbial sampler to examine the genomic blueprint of the microorganisms and a Pump Profiling System to collect high-resolution nutrient profiles in the water column. Data collected during this expedition will help create better regional models to predict what will happen to OMZs as the climate changes.
The Oxygen Minimum Zone and its Hidden Microbial World
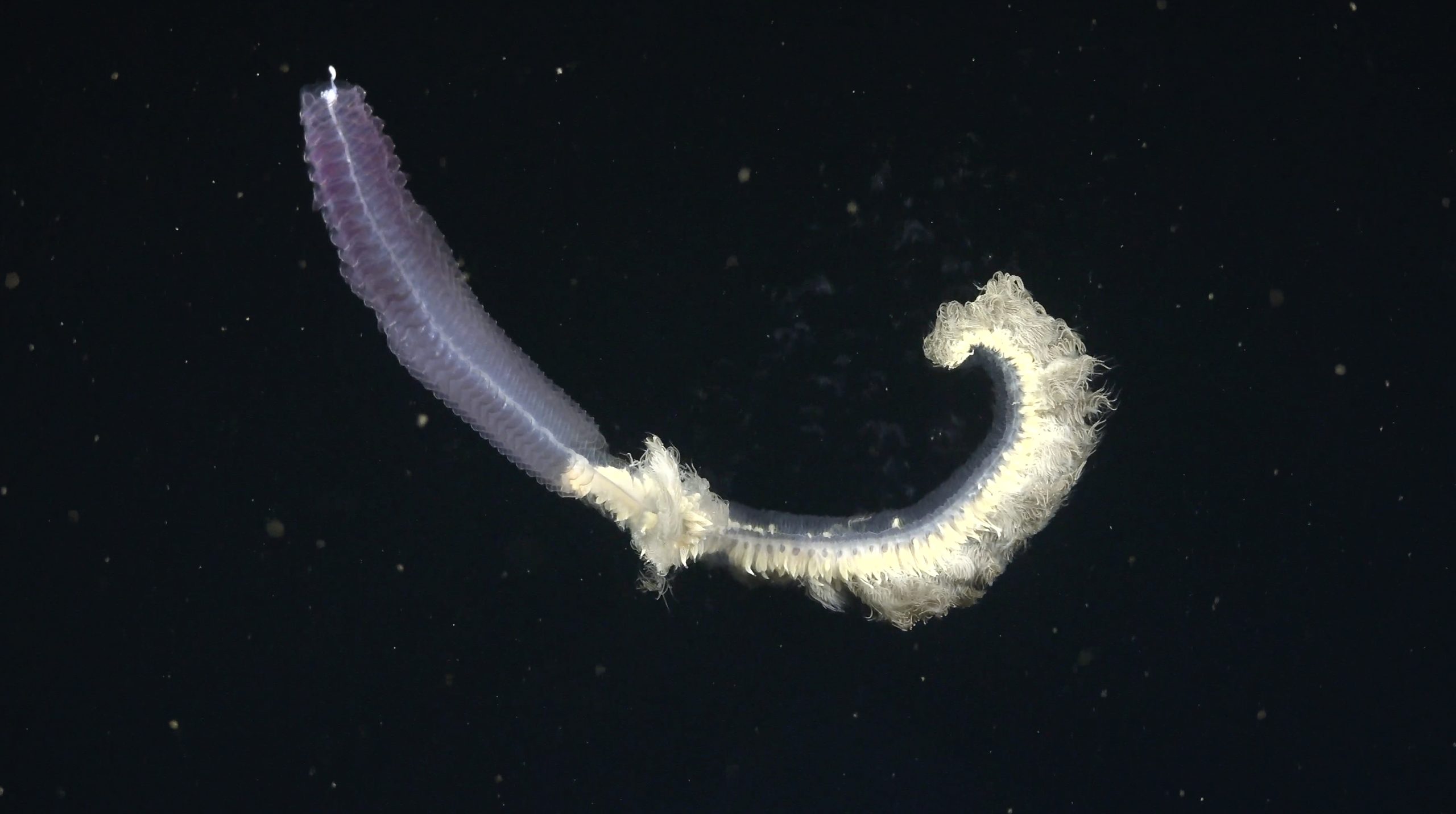
The OMZ is a naturally occurring part of the global Ocean where oxygen concentration in the water is significantly lower than average. The zone usually occurs between the depths of 100 and 1,000 meters, but this varies depending on the region. In the OMZ, the consumption of sinking organic material outstrips oxygen availability and, combined with low Ocean circulation, leaves little to no oxygen in the water column. Some large animals have evolved to live here — animals like the vampire squid that possess special adaptations for living in an oxygen-starved environment. Other oxygen-breathing animals, like sperm whales, Humboldt squid, and plankton, have adaptations for vertically migrating across the OMZ from deep to shallow water. Microbes, however, are the rulers of the OMZ and have been living in and facilitating it for millennia.
Microbes are everywhere. They thrive from the Ocean’s surface to the Mariana Trench, the deepest place on Earth. If you put every microbe and animal in the Ocean on a scale, the microbes would be 80 percent heavier. In the absence of oxygen, OMZ microbes consume molecules like ammonia, nitrite, and sulfide to gain energy, often producing gasses that have a pronounced effect on climate, such as nitrous oxide, which is 300 times more potent than CO2. OMZs are regions where several biogeochemical loops are closed in the Ocean, including the nitrogen cycle, making them a critically important part of understanding Earth’s processes.
OMZs appear to be growing due to anthropogenic climate change, potentially threatening numerous species that have not adapted to living in them. Growing OMZs may add additional greenhouse gasses, such as methane and nitrous oxide, into the atmosphere. They are also more acidic areas, further amplifying Ocean acidification. Additionally, low oxygen regions are increasing in size, number, and frequency in our Ocean’s coastal zones, with this increase linked to anthropogenic impacts such as human chemical waste, like fertilizers, entering the environment.
Scientific knowledge of microbial life in OMZs is limited and potentially flawed. Until recently, there was no instrumentation for studying these microbial processes in their natural environment, and many of the instruments used in studying OMZs result in samples becoming contaminated with oxygen before they reach the surface. Dr. Maria Pachiadaki of Woods Hole Oceanographic Institute, Dr. Laura Bristow of the University of Gothenburg, Sweden, and their teams have developed new technologies to overcome this problem and study these microbes in situ, potentially shedding light on how these microbes actually function and thrive, providing key data for revising scientific models of OMZs.
Technology for Visualizing the Invisible
Data on OMZs and the microbial communities that live in them are potentially flawed because traditional water sampling systems, like Niskin bottles, become contaminated with oxygen as they travel back to the research vessel. Oceanographic research equipment is often made from rubber and plastic, materials that easily absorb and release oxygen. Because traditional sampling methods become contaminated with oxygen before returning to the lab, historic OMZ measurements likely do not represent how microbes act in their natural environment.
To overcome these historic data limitations, Pachiadaki, Bristow, Ulloa, and their colleagues will bring a suite of instruments designed specifically for OMZs to obtain accurate measurements of microbial processes in an oxygen-starved environment.
The Pump Profiling System
The first instrument the science team will use is a Pump Profiling System, or PPS, developed by Dr. Osvaldo Ulloa from the University of Concepción in Chile. The PPS will collect real-time high-resolution vertical profile data of oxygen and nutrients in the water column, and samples of molecules like methane and other greenhouse gasses. Flow cytometry, a method for collecting cells, will be conducted utilizing the PPS to collect molecular data on the types of microbes in the water column.
The PPS is the first step in the process of describing this OMZ. The science team will take the PPS data to identify locations for deploying the Submersible Incubation Device, or SID.
Submersible Incubation Devices
The SID is a unique robotic system made from glass and titanium that has been developed through a collaboration of the teams of Pachiadaki, Bristow, and Dr Morten Larsen, and Prof Bo Thamdrup at the University of Southern Denmark. Two SIDs will be attached together and mounted on a drifting mooring, floating in the Ocean for upwards of 24 hours for each deployment.
The automated system will act like giant glass syringes, pulling water into four individual chambers. Tracers will then be added to each chamber, marking the initiation of the incubation. Microorganisms will use a tracer to produce gasses or other compounds that the team will later measure in the lab. Samples will be taken at predefined intervals without human intervention and, after recovery of the SID, will be stored and analyzed back onshore.
This instrumentation will allow the team to gain representative rates of the turnover of key elements, like nitrogen, for the first time at the exact chemical and physical conditions of the OMZ environment. Gaining representative turnover rates will facilitate the team’s understanding of how climate-relevant gasses like nitrous oxide are produced. Since the OMZ microorganisms and the processes performed are sensitive to very small changes in oxygen, the team will simultaneously monitor the oxygen conditions in each chamber.
McLane Microbial Sampler
Another device the team will deploy is a McLane Microbial Sampler. The sampler is akin to a pump and will be deployed from the aft deck. Per deployment, the sampler will collect up to 24 in situ samples of marine microorganisms and particles. The sampler will corroborate data from the PPS and SID, assessing which chemicals and microbes are in the OMZ. The sampler is essential for collecting genetic data, like RNA and DNA, from the OMZ.




Data & Publications
ADCP data collected by R/V Falkor (too) during this expedition is now available on the University of Hawaii’s Currents data page.
Environmental data collected by sensors embedded on R/V Falkor (too) during the expedition has been archived on Rolling Deck to Repository.
Radiance data collected from the HyperPro instrument has been published on Zenodo.
Processed CTD data generated from the expedition has been published on MGDS.
In the News
Photos: New tech enables marine microbes’ study in oxygen minimum zone
Interesting Engineering | May 29, 2024
New Technologies Revise Scientists’ Understanding of the Oxygen Minimum Zone
ECO Magazine| May 25, 2024
New Technologies Revise Our Understanding Of Oxygen Minimum Zones – Astrobiology
Astrobiology | May 22, 2024
DISCOVERY: New Tech Aids Understanding of the Oxygen Minimum Zone
Marine Technology News | May 21, 2024
Hacer visible lo invisible: arte y ciencia en el marEl Mostrador
El Mostrador | May 18, 2024
Team














