When we talk about the air we breathe, we often focus on oxygen. But only about 20 percent of the atmosphere consists of oxygen—the rest is almost entirely nitrogen. While nitrogen is useless to humans as a source of energy, other organisms depend on it exclusively for their survival. Tyler Tamasi, one of the members on our team, made this analogy: “If carbon is the wood, nitrogen is the nails holding everything together.”
Organisms that depend on nitrogen – like the bacteria and archaea we are studying on this expedition – cannot be seen with the naked eye. Despite their small size, an uncountable number exist on earth, and together their actions shape the planet we live on. Nitrogen forms a remarkable number of different chemical compounds, each used only by specific organisms. For example, the most abundant form of nitrogen in the atmosphere (dinitrogen gas – N2) is only available to a relatively small set of micro-organisms called nitrogen fixers. These fixers reduce N2 to ammonium (NH4+), which can then be used by different life forms. Other microbes continue the cycle, converting one nitrogen compound to another.
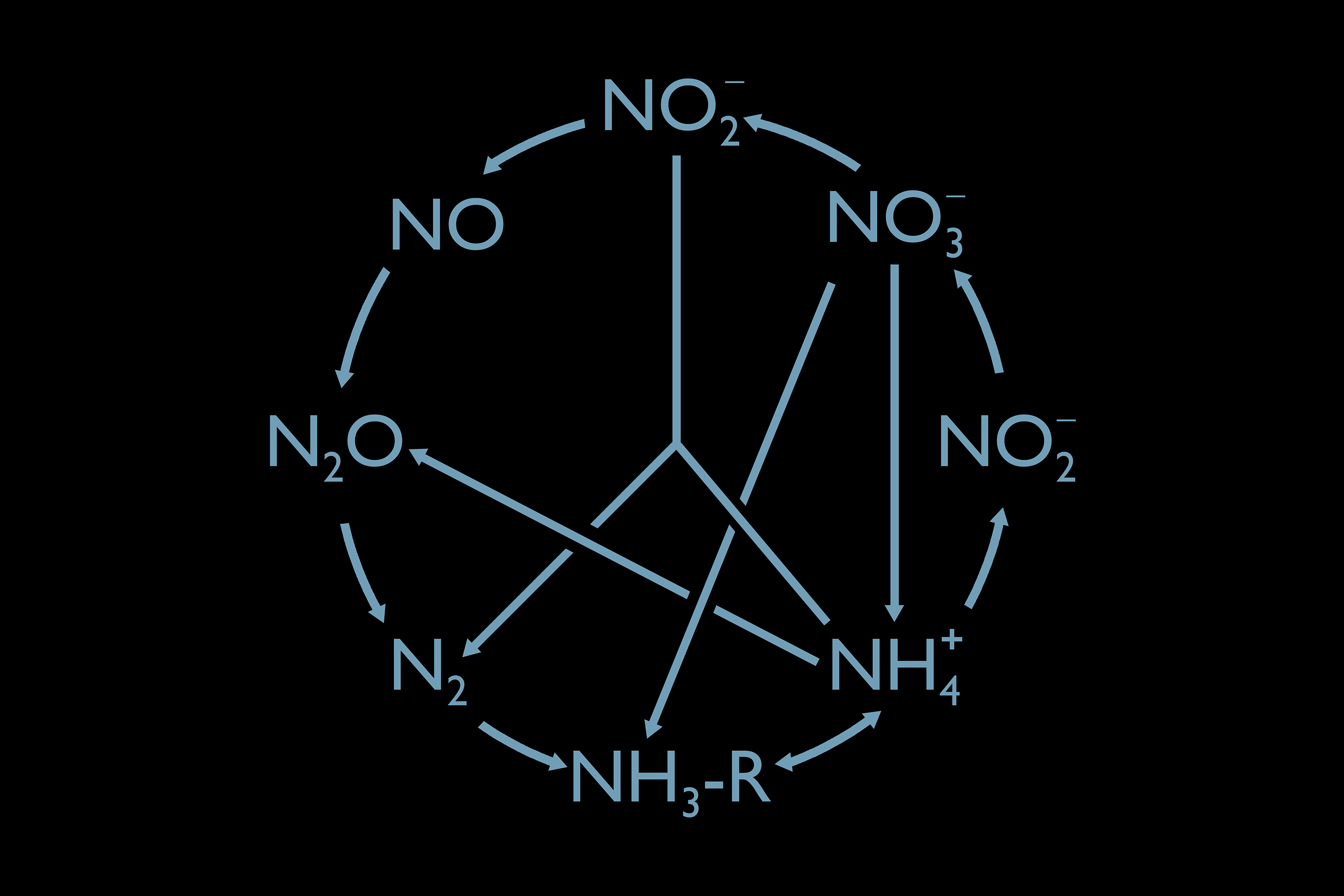
“Not all compounds are created equal,” Andrew Babbin, our chief scientist, explained in his briefing today. All life as we know it requires amino acids, which contain amines (NH2-R). Algae love nitrate (NO3–), but nitrite (NO2–) poisons fish. Nitrous oxide (N2O), commonly known as laughing gas, is one of the worst greenhouse gases in our atmosphere and the greatest ozone-depleting emission in the 21st century.
In the Zone
In the ocean, oxygen concentrations control the distribution of nitrogen compounds found in the water because different microbial processes occur faster under different oxygen conditions. Our lab (self-titled the “Bablab”) is particularly interested in looking at the microbes and processes in water where oxygen concentrations are at or near zero. We call these places oxygen minimum zones (OMZs), and they contain the only seawater where there is a net conversion of nitrogen to N2 gas. There is also evidence that OMZs produce unusually large nitrous oxide emissions to the atmosphere, making these regions critical to our understanding of climate change.
We are heading to the Eastern Tropical North Pacific, one of the three major oxygen minimum zones on Earth. As we make our way there, we are busy preparing materials and instruments to be ready to go as soon as we arrive. Yesterday, we spent the morning labeling 3,400 small vials we will use in our experiments.
In Their Native Environment
Today, we began setting up the “in-situ incubators” — innovative experimental systems that our lab is using to characterize these regions. Unlike previous experiments where ocean samples were brought back on board to study, these devices allow us to examine the microbes in their native environment without risking oxygen contamination.

At each sampling site, the ship’s crew will lower the incubators underwater and leave them suspended for a period of several hours. By adding isotopic tracers (nitrogen with 8 neutrons instead of the usual 7) to the samples, we will be able to track the dynamic changes in the levels of the samples’ different nitrogen compounds, and from these measurements, calculate the speed of each biological pathway.
These findings could be game-changing in our understanding of ocean microbes, and allow researchers to better predict how a warming climate and expansion of low oxygen “dead” zones will threaten the livelihoods of many coastal communities.

